gb GENETIC Gilbert
gb GENETIC Gilbert
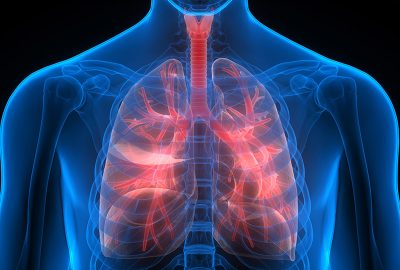
The CE IVD real-time PCR kit is intended for detection of polymorphism TA (allele 7TA) in gene UGT1A1 in human genomic DNA. Detection is based on polymerase chain reaction (PCR) with analysis of melting curves using fluorescently labelled probe.